Prurisol™ is Cellceutix’s anti-psoriasis drug candidate. It is a small molecule (MW=344) that acts through immune modulation and PRINS reduction. Prurisol has been found to be effective against psoriasis in animal models, both in induced psoriasis as well as a xenograft model with human psoriatic tissue. Prurisol is synthesized through a five-step process using commercially available starting materials.
Clinical Development
Cellceutix is developing Prurisol under FDA guidance that a 505(b)(2) designation is an appropriate development pathway. Under the FDA’s 505(b)(2) regulatory pathway, a drug’s road to market approval can be significantly shortened and at much reduced costs. Often only one pivotal Phase 3 study, enrolling a smaller number of patients than is typical, may be required. As well, the drug is eligible for up to five years of market exclusivity post-approval. For more information about the FDA’s 505(b)(2) program, please visit: http://www.fda.gov/downloads/Drugs/…/Guidances/ucm079345.pdf
Cellceutix Phase 2 Trial of Prurisol for Mild to Moderate Psoriasis Meets Primary Endpoint
- Clinical efficacy demonstrated in the highest dose (200mg) comparator arm
- Compound shown to be safe and well-tolerated with a dose-related response
- Oral delivery often preferred among patients, increasing adherence to treatment
- Additional studies planned in moderate to severe psoriasis and eczema
BEVERLY, MA–(Marketwired – May 24, 2016) - Cellceutix Corporation (OTC: CTIX) (the “Company”), a clinical stage biopharmaceutical company developing innovative therapies with oncology, dermatology, antibiotic, and anti-inflammatory applications, is pleased to inform shareholders that topline data from the Company’s Phase 2 FDA trial of orally-administered Prurisol in the treatment of mild to moderate chronic plaque psoriasis have been compiled and reviewed. The trial successfully achieved its primary endpoint, further validating Prurisol’s potential as a novel oral treatment for psoriasis.
Study Background
Enrolling 115 patients, the placebo-controlled, randomized, double-blind trial tested the efficacy and safety of three separate, twice-daily, dosing regimens of Prurisol—50 milligram (mg) (50mg QD), 100mg (50mg BD), and 200mg (100mg BD). All patients were assessed via the 5-point Investigator’s Global Assessment (IGA) scale, ranging from a score of 0 (“clear”) to a score of 4 (“severe”). The IGA scale is preferred by the U.S. Food and Drug Administration (FDA) and is comparable to the older and more commonly used Psoriasis Area and Severity Index (PASI) in evaluating psoriasis severity of patients, with many dermatologists preferring it in the clinical trial setting. Generally, an IGA score of 0/1 demonstrates a strong association with a PASI 90 score.
For more information comparing the IGA and the PASI psoriasis scoring systems, please see the link immediately below. Two additional links have been provided summarizing recent dermatological studies by Regeneron and Anacor that used the IGA scale.
- http://www.ncbi.nlm.nih.gov/pubmed/24354461
- http://newsroom.regeneron.com/releasedetail.cfm?ReleaseID=963078
- http://investor.anacor.com/releasedetail.cfm?releaseid=921668
Entry criteria for the study required: a total Body Surface Area (BSA) affected by plaque psoriasis of 10 percent to 20 percent; a baseline IGA score of 2 (“mild”) or 3 (“moderate”); and the identification of a target psoriatic lesion with a score greater than or equal to 3 based on a different (than the IGA) lesion-specific 5-point scoring scale. Clinical signs that psoriasis is clearing typically are more noticeable in patients with a greater severity of symptoms. This translated into a rigorous and aggressive study design for the Prurisol trial.
The primary endpoint assessed was the percentage of patients achieving at least a 2-point improvement from baseline on the IGA 5-point scale as measured by visual inspection of patient lesions at the end of the 84-day (12-week) treatment period. In effect, given the entry criteria, participants had to at least obtain an IGA score of “clear” or “almost clear” skin, dropping to 0 or 1 after starting from a baseline of 2 or 3. Secondary endpoints included additional improvement measures tied to degree of patient response at various time intervals.
Results Summary
The Phase 2 Prurisol trial, while not powered to demonstrate statistical significance, was conducted to inform any future fully-powered Phase 3 trial(s) that might be merited. As a result, the study’s main goal was to provide indications of efficacy, safety and tolerability upon treating patients with mild to moderate plaque psoriasis via oral delivery.
Overall analyses showed Prurisol, which is being developed under the FDA’s 505(b)(2) program, to be superior to placebo in the 200mg arm. Pharmacokinetics/Pharmacodynamics (PK/PD) further revealed an early (by week 8) dose-related response that improved as treatment duration increased.
Evaluating the primary endpoint at 84-days (week 12) in the 200mg arm, 35.0% of the patients receiving that dose of Prurisol demonstrated clinically significant improvements compared with 16.7% of patients on placebo only. This percentage includes patient data from one site where investigator non-compliance may have occurred. Were that site to have been excluded from overall data analysis, as is done in some clinical studies (refer to the journal article linked to below, published findings from another psoriasis study), 43.7% of patients in the 200mg Prurisol arm would have met the primary endpoint. Patient responses in the 50mg and 100mg arms were statistically comparable to the placebo arm.
For purposes of direct comparison, the Prurisol trial outperformed a similarly designed Phase 2b trial in the treatment of mild to moderate psoriasis conducted in 2011 by Anacor Pharmaceuticals in which a topical anti-inflammatory compound was assessed. See the link below.
Sub-population analyses further showed greater efficacy demonstrated in patients who had a baseline IGA score of 3 (“moderate”) as compared to those with a baseline score of 2 (“mild”). Some of these patients even experienced a 3-point reduction in their IGA score, going from “moderate” to “clear.” This suggests Prurisol may be more effective in treating moderate to severe psoriasis patients to a greater degree than those patients who exhibit less severe symptoms. In moderate to severe psoriasis studies, the placebo response also tends to be lower.
Regarding Prurisol’s safety profile, only a single Serious Adverse Event (SAE) was reported in the study, that being in the 50mg arm, with the type and the rate of occurrence of additional Adverse Events (AEs) similar and evenly distributed across all the three dosing arms and the placebo arm.
Additional detailed data review and analysis is underway and an end-of-Phase 2 meeting with the FDA will be requested to help determine the dose(s) and design for future studies, which may include higher dosing regimens to determine Prurisol’s maximum therapeutic effect. The Company plans to release other summary findings from this trial in the coming months, with full results anticipated to be submitted for journal publication and presentation at a future medical congress.
Investigator Comments
Cellceutix would also like to share some observations and comments from Principle Investigators (PIs) overseeing different participating clinical sites in the trial. Numerous PIs noted patients expressed a desire to have access to Prurisol following the study’s conclusion. Moreover, the Company learned that some patients were previously unsuccessfully treated with other therapies, including biologics and apremilast (Otezla®).
Included below is a sampling of responses that were authorized to be published:
- “Well designed study, patients were pleased, minimal to no side effects.”
- “Good tolerability was shown with very few AEs reported. Good compliance and good patient satisfaction.”
- “Overall impression of the study is very positive, as well as the patients’ satisfaction.”
When asked, “Why would you choose Prurisol (should it be approved) over another oral compound for psoriasis?” and “Why would you choose Prurisol over an injectable biologic?” responses included:
- “Because [Prurisol] does not cause any severe side effects. Patients dislike needles and injections because they cause pain and discomfort.”
- “Patients overall prefer oral medications over injectable medication. Our experience with injectable studies for other indications has been that many subjects do not like taking them and those studies tend to be difficult compared to oral medications to recruit subjects.”
- “Because of [Prurisol’s] low side effect profile.”
- “Patients prefer oral treatments.”
Sponsor Comments
Cellceutix believes that the results from the Prurisol Phase 2 trial are extremely encouraging, especially for the 80 percent of psoriasis sufferers exhibiting milder symptoms of the condition. These people are more likely to switch between therapies or be off treatment altogether. Planning is underway to explore Prurisol’s potential in the treatment of moderate to severe psoriasis and eczema, a skin condition experienced by up to 30 percent of children and 10 percent of adults. The Company is hopeful Prurisol may one day become a leading treatment for psoriasis regardless of disease severity.
Leo Ehrlich, Chief Executive Officer of Cellceutix, commented: “We had always wanted to explore Prurisol’s clinical merit, as it had excellent results in laboratory studies. We put it to the test under some of the most demanding conditions, with respect to the IGA versus PASI scoring systems; short treatment duration; low dosing levels; enrollment that included patients who were previously treated with biologics; and evaluation in mild to moderate psoriasis patients, where it can be more difficult to achieve a meaningful therapeutic effect. To see such a strong response among patients, achieving clear to almost clear skin without serious side effects—the downside of biologics—in such a short period of time, is exceptional. We would like to thank patients and investigators who participated in the study. Prurisol has taken a key step towards potentially becoming only the second oral treatment approved by the FDA for psoriasis in decades.”
Leo Ehrlich continued: “More broadly, these outstanding results are the first of what we hope are more to come as the Cellceutix pipeline continues to progress. With our three lead drugs, Prurisol, Brilacidin, and Kevetrin, now in later-stage FDA trials, each a possible first-in-class treatment, we are thrilled with what the future holds.”
About Psoriasis
Affecting an estimated 125 million people worldwide, psoriasis is a chronic immune-mediated skin disorder presenting with varying symptoms and levels of severity. The condition is characterized by raised and inflamed patches of skin, often on the elbows, knees, scalp, hands and feet, and causes itching, irritation, stinging and pain. Often feeling socially stigmatized, over 80 percent of people with psoriasis report it negatively impacts the quality of their everyday life. Mild cases are defined as affecting less than 3 percent of the body’s surface area, with a majority of cases limited to less than 2 percent of the skin. Moderate psoriasis covers 3 to 10 percent of the skin. If it covers 10 percent of the body, the disease is considered severe. Up to 40 percent of psoriasis patients will develop psoriatic arthritis (PsA) within 7 to 12 years. Psoriasis also is associated with numerous comorbidities, ranging from cardiovascular disease, autoimmune disease, and cancer to psychological disorders. Despite recent advances, there remains a need for orally-delivered psoriasis drugs, and other treatment alternatives to biologics, which are known to have serious side effects and contraindications, and generally lose their effectiveness over time. Additional information can be found by reading the World Health Organization’s (WHO) 2016 Global Report on Psoriasis: http://apps.who.int/iris/bitstream/10665/204417/1/9789241565189_eng.pdf
Prurisol Labratory Summary
Prurisol is more effective than conventional therapies in human psoriatic tissue xenografts
» Prurisol (10 mg/kg PO twice/ day x 21 days)
» 84% reduction in lesion appearance
» 99% reduction in lesions based on histology
» 96% reduction in serum PRINS
» 87% reduction in serum IL-20
» No reoccurrence of lesions within 180 days
» Methotrexate (7.5 mg/kg IP once/ day x 5 days)
» 63% reduction in lesion appearance
» 76% reduction in lesions based on histology
» 48% reduction in serum PRINS
» 46% reduction in serum IL-20
» Lesions reoccurred in 61 days
Prurisol is a small molecule, acting on the principles of immune modulation and PRINS reduction that has been found to be effective against psoriasis in animal models, both in induced psoriasis as well as a xenograft model with human psoriatic tissue.
Details
Prurisol was studied in mice that were irradiated with 350 Rads whole body to suppress graft rejection, and then engrafted with human psoriatic tissue by inserting human psoriatic tissue under the skin using a trocar. Groups of ten mice were treated with Prurisol orally for 21 days with either 10 mg/kg Prurisol once/ day or 10 mg/kg Prurisol twice/day, or with 7.5 mg/kg methotrexate IP once/day, or untreated mice as controls. The mice were followed for 180 days. Endpoints were skin appearance, based on a score of 0 for normal appearance to 10 for severe lesion, histological observations, also based on a score of 0 for normal appearance to 10 for severe lesion, PRINS RNA in psoriatic lesions, and blood IL-20 levels. For these parameters, Prurisol was compared to controls and methotrexate.
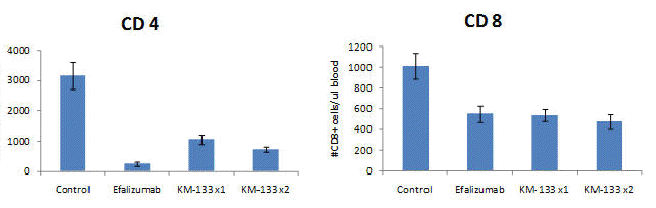
In a second experiment, groups of 10 immunocompetent CD-1 mice were treated with one or two doses of 10 mg/kg Prurisol daily or 3 mg/kg efalizumab SC once per week for 3 weeks. CD4+ and CD8+ lymphocyte counts were also measured by flow cytometry and compared to efalizumab. Prurisol significantly reduced all psoriatic endpoints measured relative to controls

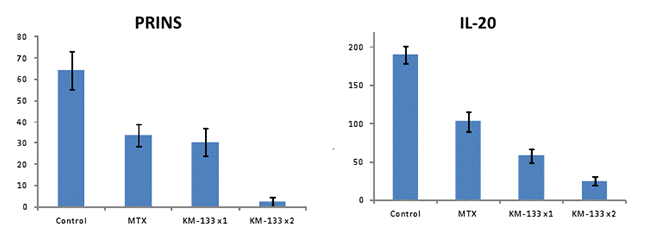
In addition, psoriasis did not recur during the study period with the higher dose of Prurisol whereas with methotrexate, psoriasis recurred after an average of 61 days. Treatment with Prurisol caused less reduction in CD4+ lymphocyte counts than did efalizumab. Weight loss in the treated animals was within acceptable limits.
Prurisol given at 10 mg/kg twice/day was shown to be more effective than methotrexate in reducing psoriatic skin lesions in a human xenograft model. In addition, Prurisol can be given orally and is well-tolerated.
A variation of Prurisol in human xenograft model. The top row animals show a relatively clean coat with limited discernibility of psoriasis. The bottom row are the untreated control animals.
By SAR, the lead candidate Prurisol is selected. Prurisol in human xenograft model. The top row animals show a clean coat with no evidence of psoriasis, essentially showing that Prurisol cured the psoriasis in the mice. The bottom row shows the untreated control animals.
